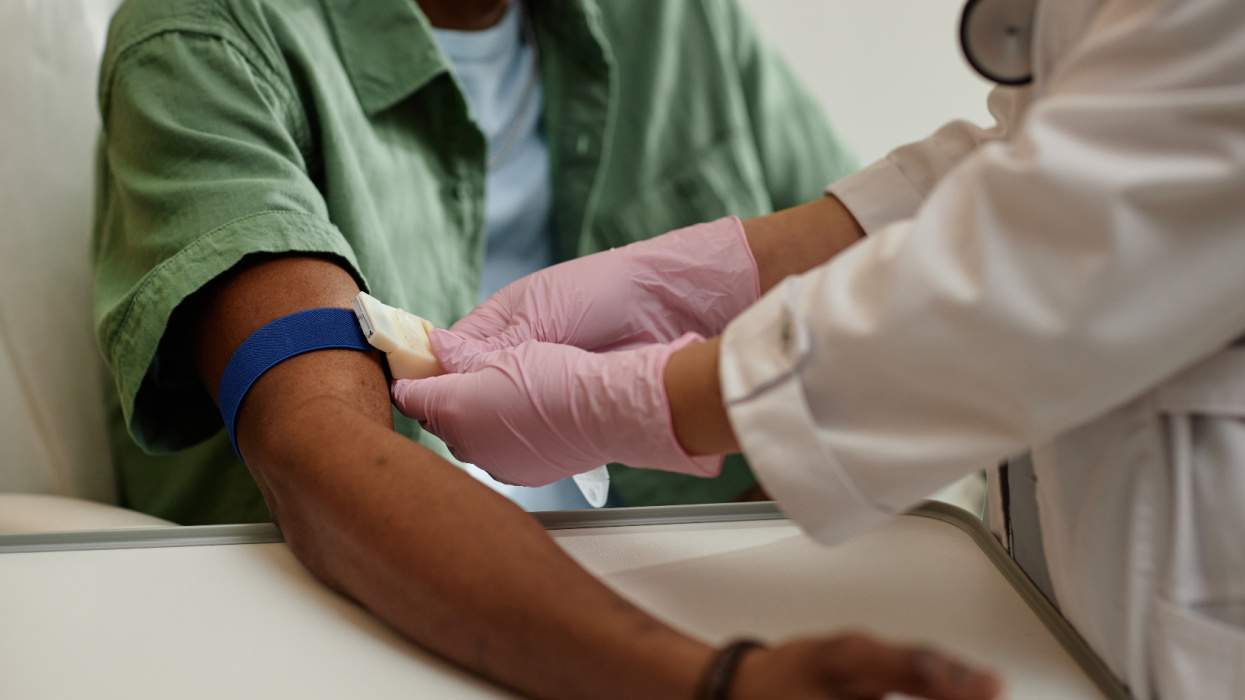
A panel of experts in Paris on Monday released international guidelines for treating HIV-positive adults with the HIV fusion inhibitor Fuzeon. The nine-member panel called for intensified training of health care workers and HIV patients on how to properly inject the medication, including regular follow-up assessment visits. The panel also recommended that Fuzeon be given to patients with CD4-cell counts of at least 100 whenever possible and that the medication be dosed while a patient is still responsive to at least two other anti-HIV drugs. "For early-stage patients, the objective of therapy is for the patient to achieve an undetectable level of HIV," the panel said in a press release. "For later-stage patients, however, the focus should shift toward achieving a boosted immune system (CD4 count) rather than focusing exclusively on a drop in HIV levels." Fuzeon is a synthetic peptide that works by preventing HIV from attaching to and infecting immune system cells. Clinical trials of the drug showed the medication boosted CD4-cell counts and lowered HIV viral loads in patients who had developed resistance to protease inhibitors. The drug, which is priced at about $20,000 for a one-year supply, making it the most expensive anti-HIV drug on the market, is jointly marketed by Roche and Trimeris.
Search
AI Powered
Human content,
AI powered search.
Latest Stories
Stay up to date with the latest in LGBTQ+ news with The Advocate’s email newsletter, in your inbox five days a week.
@ 2026 Equal Entertainment LLC.
All rights reserved
All rights reserved
By continuing to use our site, you agree to our Privacy Policy and Terms of Use.
The Latest
Support Independent Journalism
LGBTQ+ stories deserve to betold.
Your membership powers The Advocate's original reporting—stories that inform, protect, and celebrate our community.
Become a Member
FOR AS LITTLE AS $5. CANCEL ANYTIME.
More For You
Most Popular
@ 2026 Equal Entertainment LLC. All Rights reserved