In private, the company that manufactures the only monkeypox vaccine approved by the Food and Drug Administration expressed concerns to the Biden administration about the FDA's plans to dilute a vial's dose into five doses and change the way the shot is administered.
The CEO of Bavarian Nordic, Paul Chaplin, wrote of his concerns to the Department of Health and Human Services in a letter obtained by the Washington Post Tuesday, the paper reports.
"Bavarian Nordic...is dedicated to assisting Governments around the globe to control the current monkeypox outbreak and is fully supportive of dose-sparing approaches, such as delaying the second vaccination," Chaplin wrote. "However, we do have some reservations on the [new injection] approach, due to the very limited safety data available..., the higher [rates of expected adverse reactions] compared to the JYNNEOS standard dose and route..., and the fact that there was a relatively high percentage of subjects...that failed to receive the second vaccination during a controlled clinical study."
The letter continues, "We have been made aware of some additional analysis of the [intradermal] study data, but essentially this does not change the overall picture that the [intradermal] administration results in increased [adverse reactions] compared to [the current route of administration] and this may have a negative impact on vaccine uptake and coverage."
For the manufacturer to present valid data concerning its product and untested protocols, Chaplin urges the administration to gather data from the rollout of the government's new dosage and administration option.
"It is our understanding that it is the United States Government position that there is no time to prepare or conduct such a protocol," Chaplin writes. "Therefore, as the manufacturer of JYNNEOS, we have already begun discussions with investigators on the feasibility of how to conduct a study to provide more safety information to help govern future vaccination policies."
Biden administration officials said they were aware of Bavarian Nordic's concerns, but that they would not change the vaccine strategy, according to the Post.
"We've had conversations with them about this, and so has FDA," Xavier Becerra, the U.S. Secretary of the Department of Health and Human Services, said. "We wouldn't have moved forward unless we thought it was safe and effective, and if FDA hadn't dotted its I's and crossed its T's."
Get the latest updates about monkeypox. Click here.
The Post also reports that some in the administration feel that the company's worries are actually from concerns that it will suffer from a loss in profit if countries extend their current supply.
A representative for Bavarian Nordic told the paper that the company's concerns were over safety.
As part of a plan to extend the nation's limited monkeypox vaccine supply, the government said Tuesday it would convert hundreds of thousands of doses of Bavarian Nordic's Jynneos vaccine into millions of potential shots to protect Americans from MPV.
Representatives from the Infectious Diseases Society of America welcomed the new administration route at a press conference Thursday morning.
However, Dr. Kevin Ard, medical director of the National LGBTQIA+ Health Education Center and director of the Massachusetts General Hospital Sexual Health Clinic, who noted that the focus should also be fighting stigma, warned that proper implementation was necessary. Not only are special needles required, but people also need to be trained.
"There is some groundwork that needs to be laid for this transition to intradermal dosing not just for supplies, but also ensuring everything is in place to comply with the EUA requirements, that we have staff who are trained in giving the vaccine, and so forth," Ard said.
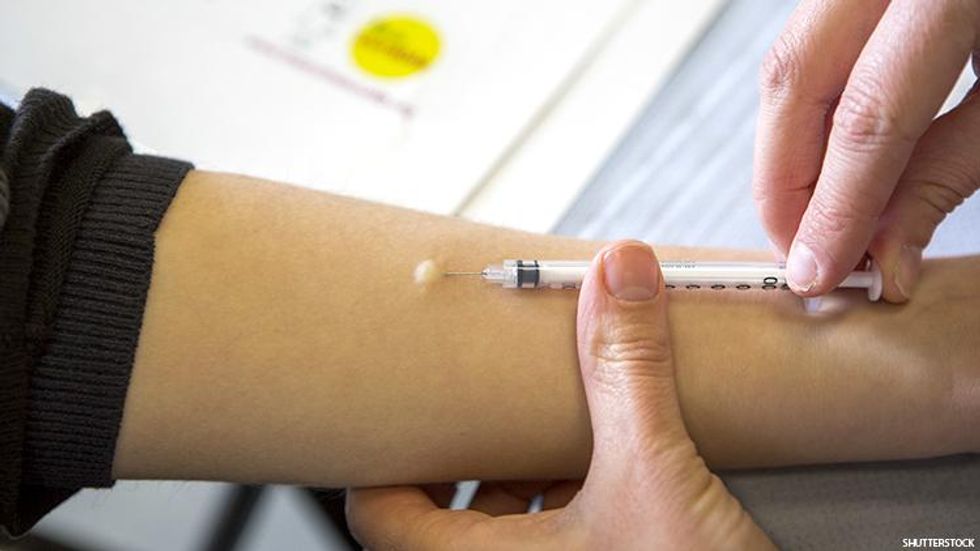
Most vaccines are delivered subcutaneously, into the fat portion of the skin, or intramuscularly, deeply into the muscles. Intradermal injection involves placing a much finer needle between the top layers of the skin, which causes a small bubble to appear under the skin. This is similar to the PPD test one might recognize from tuberculosis testing.
Above: A person receives an intradermal injection with a fine needle just between skin layers.