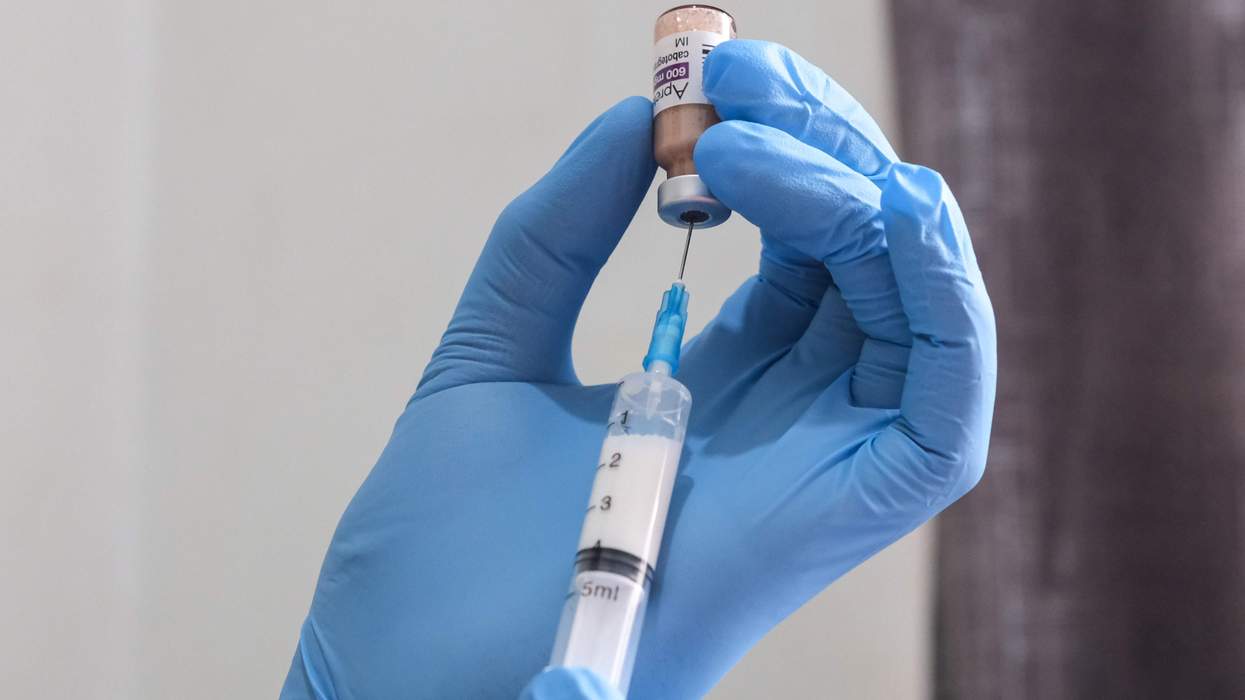
The laboratory at Maryland General Hospital--where faulty lab equipment produced about 450 suspect HIV and hepatitis tests from June 2002 through August 2003--has taken corrective action and is now in compliance with Medicare regulations, according to federal regulators. Earlier this year workers had sent out HIV and hepatitis test results although equipment indicated the results might have been inaccurate. The lab could have lost its ability to collect Medicare payments if it had not corrected the problems that lab workers had tipped off to authorities. The lab also faced a state Medicaid fraud investigation, subpoenas from the U.S. Health and Human Services' Office of the Inspector General, and congressional hearings. In response, the University of Maryland Medical System hospital brought in an outside consultant to run the lab and hired more lab workers. Colene Daniel replaced Timothy D. Miller, who resigned, as hospital CEO. All who received the suspect test results were offered retesting. And the retests confirmed that the originals were overwhelmingly accurate, the hospital says. Despite some violations, Maryland General's lab is back in compliance with Medicare's conditions, according to an August 20 letter from the federal Centers for Medicare and Medicaid, reports the Baltimore Sun. The federal agency ensures that hospitals and laboratories are properly inspected and eligible for Medicare and Medicaid payments. (AP)
Search
AI Powered
Human content,
AI powered search.
Latest Stories
Stay up to date with the latest in LGBTQ+ news with The Advocate’s email newsletter, in your inbox five days a week.
@ 2026 Equal Entertainment LLC.
All rights reserved
All rights reserved
By continuing to use our site, you agree to our Privacy Policy and Terms of Use.
The Latest
Support Independent Journalism
LGBTQ+ stories deserve to betold.
Your membership powers The Advocate's original reporting—stories that inform, protect, and celebrate our community.
Become a Member
FOR AS LITTLE AS $5. CANCEL ANYTIME.
More For You
Most Popular
@ 2026 Equal Entertainment LLC. All Rights reserved