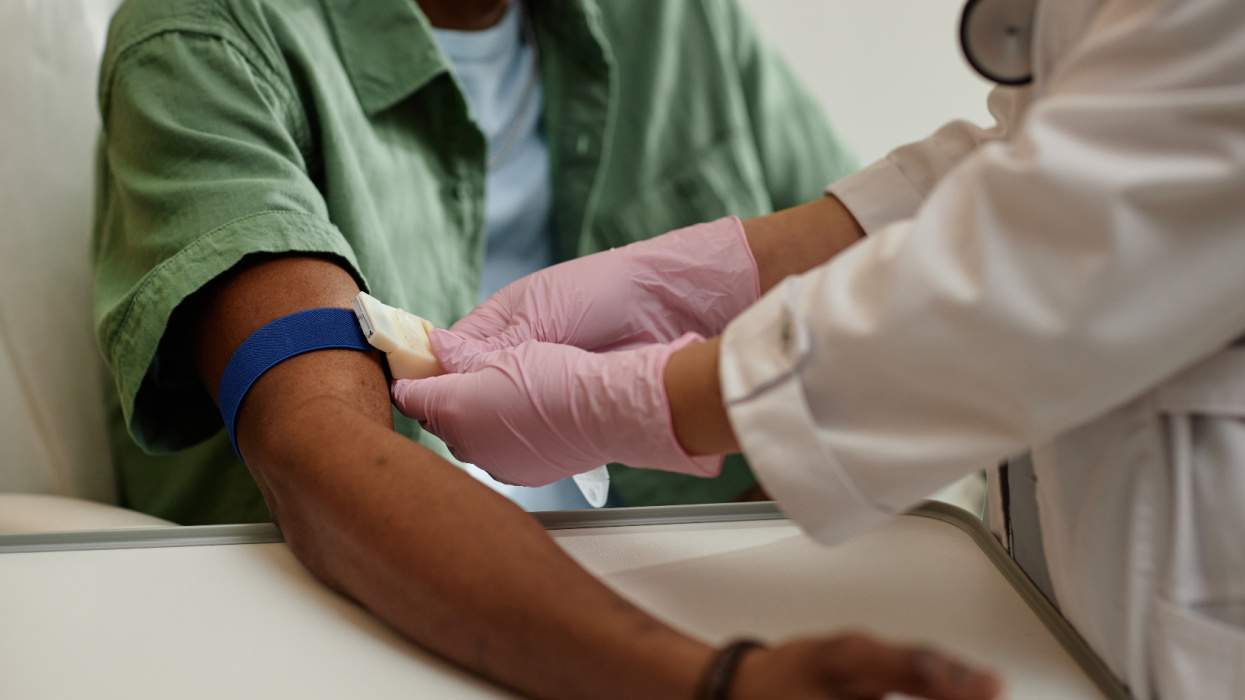
The Food and Drug Administration last week approved a new formulation of the protease inhibitor Invirase made by Roche. The new 500-milligram tablets will decrease the pill burden for HIV patients from 10 pills to four pills daily. The tablets also are smaller in size than the 200-milligram capsules currently available. "With the new Invirase 500-milligram tablet now available, pill burden is considerably reduced, and we anticipate that this improved convenience, together with 'boosted' Invirase's record of convincing data, will result in an expansion of its use in the earlier stages of HIV therapy," said William Burns, head of Roche's pharmaceutical division, in a press statement.
Search
AI Powered
Human content,
AI powered search.
Latest Stories
Stay up to date with the latest in LGBTQ+ news with The Advocate’s email newsletter, in your inbox five days a week.
@ 2026 Equal Entertainment LLC.
All rights reserved
All rights reserved
By continuing to use our site, you agree to our Privacy Policy and Terms of Use.
The Latest
Support Independent Journalism
LGBTQ+ stories deserve to betold.
Your membership powers The Advocate's original reporting—stories that inform, protect, and celebrate our community.
Become a Member
FOR AS LITTLE AS $5. CANCEL ANYTIME.
More For You
Most Popular
@ 2026 Equal Entertainment LLC. All Rights reserved