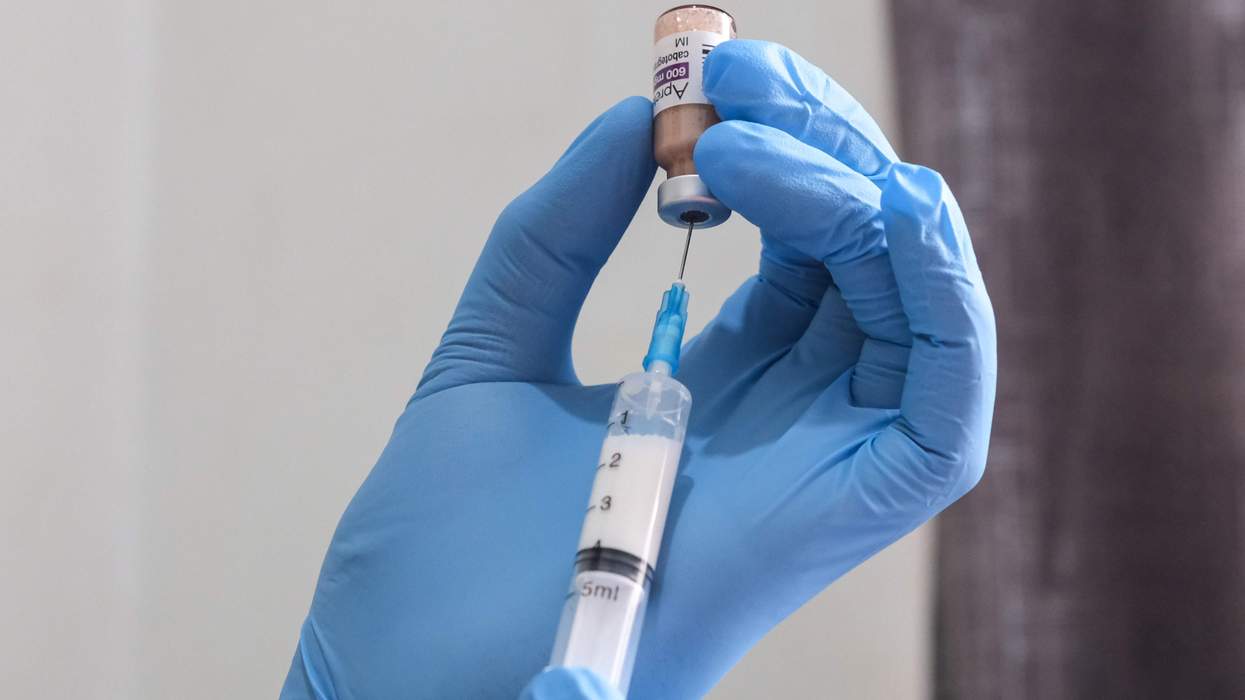
Two generic versions of a major anti-HIV drug were tentatively approved by the Food and Drug Administration on Monday. The agency said it was giving tentative approval to applications for nevirapine tablets manufactured by two companies in India: Ranbaxy Laboratories Limited in Guragon and Aurobindo Pharma in Hyderabad. These are the first generic versions of Viramune Tablets manufactured by Boehringer Ingelheim. The new generic versions won't be available in the United States because of patent exclusivity rules. But the approval means the generic versions meet the FDA's standards and can be used by relief organizations outside the United States funded by President Bush's $15 billion AIDS relief plan. Nevirapine is in the class of drugs that help keep HIV from reproducing. It is used in combination with other antiretroviral agents. The Administration was criticized last year for refusing to purchase drugs that hadn't been reviewed by the FDA. AIDS activists contended that the Administration was trying to bolster the sales of U.S. drugmakers at the expense of AIDS patients in the developing world. Research on nevirapine has drawn controversy, and in April an expert scientific panel at the Institute of Medicine concluded that while U.S. studies on the drug in Africa violated federal patient protection rules, the work was conducted well enough to support its conclusions that the drug could be used safely to protect babies. The Associated Press reported in December that the U.S. Office of Human Research Protections had concluded that the National Institutes of Health experiment in Uganda that dated to the mid 1990s had violated federal patient safety rules. (AP)
Search
AI Powered
Human content,
AI powered search.
Latest Stories
Stay up to date with the latest in LGBTQ+ news with The Advocate’s email newsletter, in your inbox five days a week.
@ 2026 Equal Entertainment LLC.
All rights reserved
All rights reserved
By continuing to use our site, you agree to our Privacy Policy and Terms of Use.
The Latest
Support Independent Journalism
LGBTQ+ stories deserve to betold.
Your membership powers The Advocate's original reporting—stories that inform, protect, and celebrate our community.
Become a Member
FOR AS LITTLE AS $5. CANCEL ANYTIME.
More For You
Most Popular
@ 2026 Equal Entertainment LLC. All Rights reserved