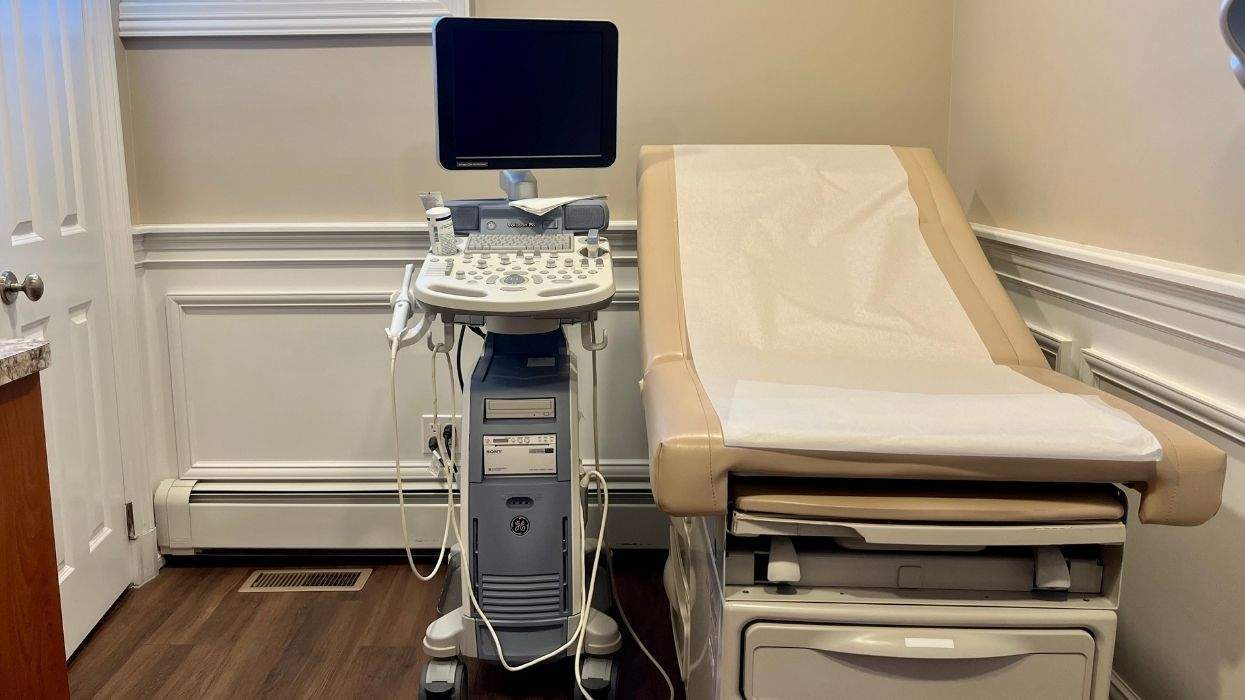
OraSure Technologies on Wednesday announced the upcoming nationwide launch of its OraQuick Advance Rapid HIV-1/2 Antibody Test, the only Food and Drug Administration-approved rapid HIV test that can detect antibodies to both HIV-1 and HIV-2 viral strains in about 20 minutes. The test can screen for HIV antibodies from oral fluids, finger-stick blood samples, whole blood, or plasma samples. It is the only rapid HIV antibody test available that can be used with oral fluid samples. Shipments of the new test to hospitals, clinics, and AIDS service groups offering HIV testing will begin in early November. The OraQuick HIV 1/2 test received FDA approval in June and subsequently received a federal waiver to allow its use at more than 180,000 U.S. sites, including physicians' offices and community-based AIDS and health groups. HIV-1 is the most common subtype of the virus in North America and Europe. HIV-2 is rare in the United States but is the predominant form of the virus in West Africa.
Search
AI Powered
Human content,
AI powered search.
Latest Stories
Stay up to date with the latest in LGBTQ+ news with The Advocate’s email newsletter, in your inbox five days a week.
@ 2026 Equal Entertainment LLC.
All rights reserved
All rights reserved
By continuing to use our site, you agree to our Privacy Policy and Terms of Use.
The Latest
Support Independent Journalism
LGBTQ+ stories deserve to betold.
Your membership powers The Advocate's original reporting—stories that inform, protect, and celebrate our community.
Become a Member
FOR AS LITTLE AS $5. CANCEL ANYTIME.
More For You
Most Popular
@ 2026 Equal Entertainment LLC. All Rights reserved