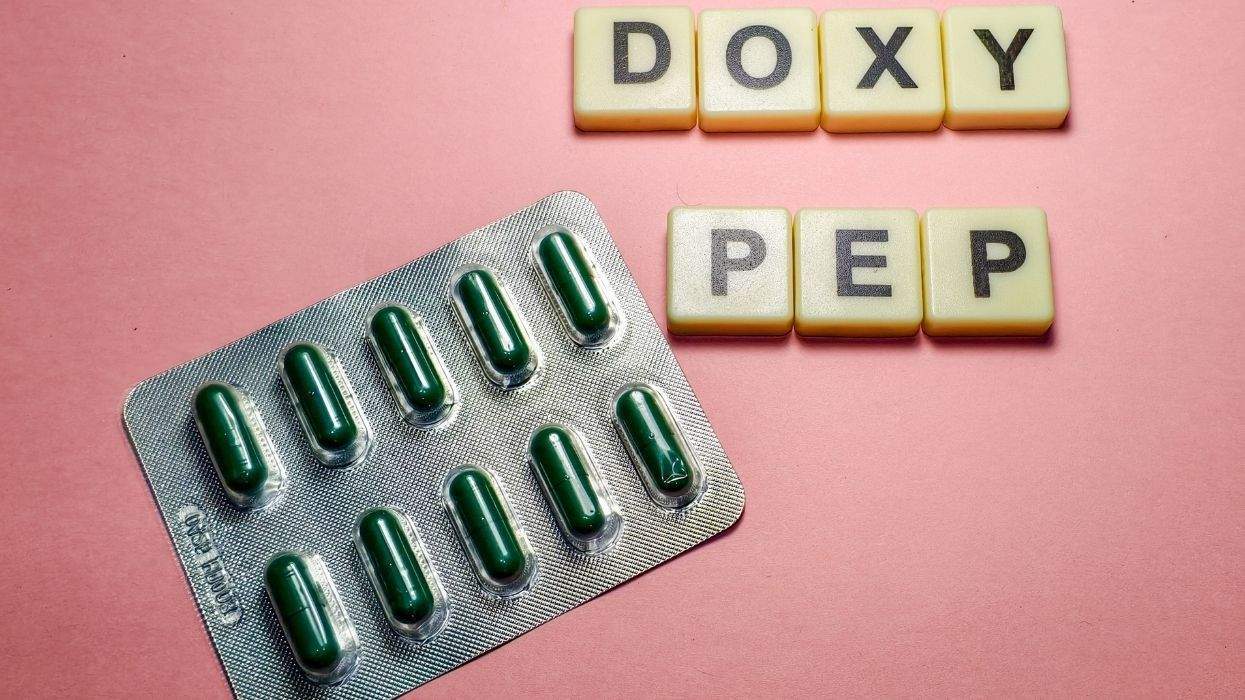
AIDS activists in Bangkok, Thailand, are protesting plans, proposed by the U.S. Centers for Disease Control and Prevention and approved by Thailand's health ministry, to test the efficacy of the anti-HIV drug Viread in preventing HIV infections in hundreds of volunteers. The Thai trial will involve 1,600 intravenous-drug users in Bangkok. The drug is also to be tested as preexposure prophylaxis at sites in the United States and Africa. In some countries planned trials of Viread have been canceled amid allegations that its manufacturer, Gilead Sciences, failed to take steps to prevent subjects from being infected and did not offer continuing care for those who might contract HIV. In August, Cambodia's premier halted a Viread trial that would have involved hundreds of sex workers. A planned trial in Cameroon was temporarily halted earlier this year. The Thai Drug Users' Network and other AIDS groups issued a statement last week opposing Thailand's approval of the tests because of alleged ethical violations and the failure to consult the entire HIV community. Paisan Suwannawong, director of the Thai AIDS Treatment Action Group, alleged that Gilead Sciences has failed to provide subjects with injecting equipment. Seree Jintakanon, chair of the Thai Drug Users' Network, said there has been no promise of treatment for volunteers diagnosed with HIV when seeking to join the trial or for those who might become infected during the three-year test. Last year Gilead responded to such concerns by saying all participants receive extensive counseling. The company said it is not compelled to provide lifetime health coverage for participants because they are being given prevention information that will make them less likely to contract HIV. (AP)
Search
AI Powered
Human content,
AI powered search.
Latest Stories
Stay up to date with the latest in LGBTQ+ news with The Advocate’s email newsletter, in your inbox five days a week.
@ 2026 Equal Entertainment LLC.
All rights reserved
All rights reserved
By continuing to use our site, you agree to our Privacy Policy and Terms of Use.
The Latest
Support Independent Journalism
LGBTQ+ stories deserve to betold.
Your membership powers The Advocate's original reporting—stories that inform, protect, and celebrate our community.
Become a Member
FOR AS LITTLE AS $5. CANCEL ANYTIME.
More For You
Most Popular
@ 2026 Equal Entertainment LLC. All Rights reserved