Brazil is still considering whether it will break patents on three anti-HIV drugs after a deadline under its royalty agreement with three U.S. drug firms lapsed April 4, says health ministry spokesman Cesar Rocha. The royalty agreement allowed Brazil to manufacture copies of Abbott Laboratories' combination lopinavir-ritonavir pill, Merck's efavirenz, and Gilead Sciences' tenofovir. "The ministry will announce its decision soon," says Rocha. "There is no defined date," he notes, denying reports in local press that the decision would be made after President Luiz Inacio da Silva returned from a trip to Africa on April 15. Brazil's intellectual property law allows the government to break patents in cases of a health emergency or if it finds a pharmaceutical company engages in abusive pricing. The government has extracted several price breaks from pharmaceutical companies by threatening such a move. For instance, Merck has reduced efavirenz's price four times. Still, it has not been enough to offset rising costs, the government says. The drugs in question will cost Brazil $169 million this year, says Rocha. That accounts for 67% of the government's budget for imported antiretroviral drugs. One company declined to grant Brazil voluntary licensing, which allows Brazil to pay the company royalties on domestically produced generics, Rocha says. Another firm's agreement foundered on conditions the ministry could not accept, and the third company has not made a decision and is still in talks, says Rocha. Ministry policy is not to name the companies or negotiating stances, he notes. No one from the three drug firms was available for immediate comment. (AP)
Search
AI Powered
Human content,
AI powered search.
Latest Stories
Stay up to date with the latest in LGBTQ+ news with The Advocate’s email newsletter, in your inbox five days a week.
@ 2026 Equal Entertainment LLC.
All rights reserved
All rights reserved
By continuing to use our site, you agree to our Privacy Policy and Terms of Use.
The Latest
Support Independent Journalism
LGBTQ+ stories deserve to betold.
Your membership powers The Advocate's original reporting—stories that inform, protect, and celebrate our community.
Become a Member
FOR AS LITTLE AS $5. CANCEL ANYTIME.
More For You
Most Popular
@ 2026 Equal Entertainment LLC. All Rights reserved













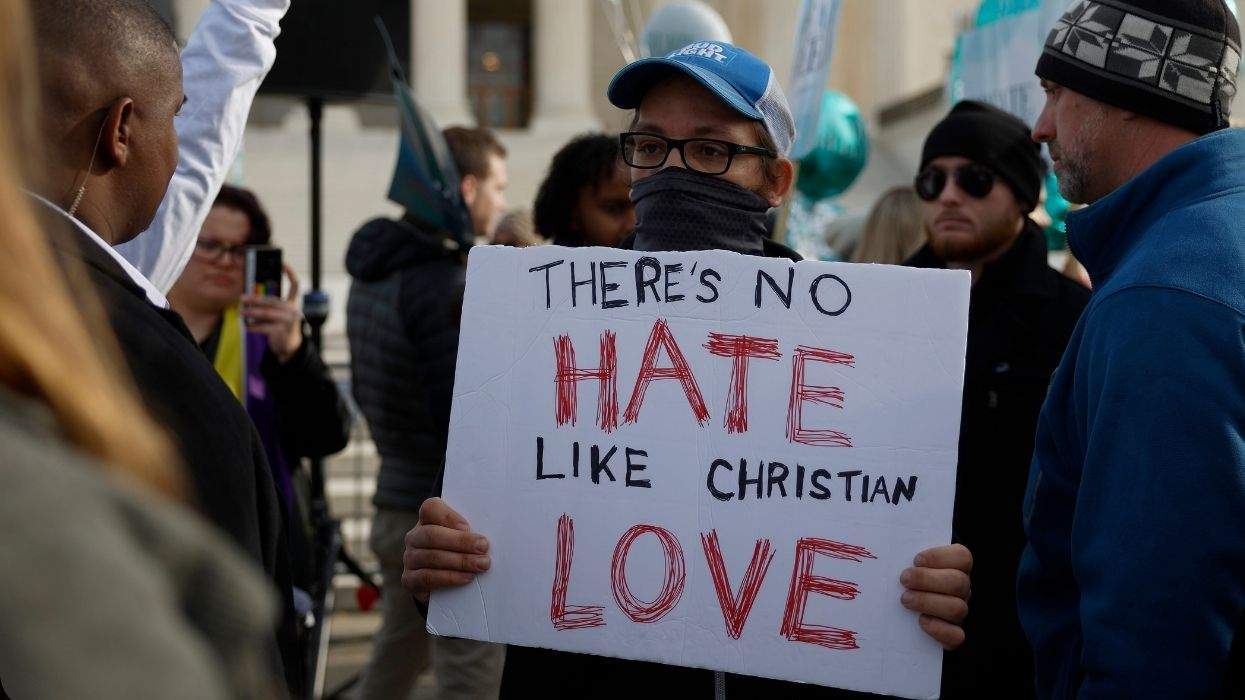
Charlie Kirk DID say stoning gay people was the 'perfect law' — and these other heinous quotes
These are some of his worst comments about LGBTQ+ people made by Charlie Kirk.