CONTACTAbout UsCAREER OPPORTUNITIESADVERTISE WITH USPRIVACY POLICYPRIVACY PREFERENCESTERMS OF USELEGAL NOTICE
© 2025 Equal Entertainment LLC.
All Rights reserved
All Rights reserved
By continuing to use our site, you agree to our Privacy Policy and Terms of Use.
We need your help
Your support makes The Advocate's original LGBTQ+ reporting possible. Become a member today to help us continue this work.
Your support makes The Advocate's original LGBTQ+ reporting possible. Become a member today to help us continue this work.
The Food and Drug Administration on Friday gave full approval for the marketing of Serostim for the treatment of HIV-related wasting. The compound received accelerated approval in 1996, a special regulatory status granted by the FDA for a drug used to treat patients with life-threatening illnesses. The full approval was based on a multi-center, placebo-controlled study of 757 HIV-positive people showing that the injectable medication is effective in increasing lean muscle mass and boosting weight in patients suffering from AIDS-related wasting. "We are very pleased that the FDA has granted full approval for Serostim," said James Sapirstein, executive vice president at Serono, the maker of the drug. "Wasting continues to be a major concern in the management of HIV and AIDS treatment, even with the benefits of highly active antiretroviral therapy. Serostim makes an important and positive difference in the physical endurance of people experiencing HIV wasting." Serostim is the only growth hormone approved by the FDA for the treatment of AIDS-related wasting. The drug is not available in Europe; in April 2003, the European Medicines Evaluation Agency rejected Serono's application to market the medication. About 6,000 HIV-positive people in the United States are prescribed Serostim. A 12-week course of the drug costs about $21,000.
From our Sponsors
Most Popular
Bizarre Epstein files reference to Trump, Putin, and oral sex with ‘Bubba’ draws scrutiny in Congress
November 14 2025 4:08 PM
True
Jeffrey Epstein’s brother says the ‘Bubba’ mentioned in Trump oral sex email is not Bill Clinton
November 16 2025 9:15 AM
True
Watch Now: Pride Today
Latest Stories
There’s a testosterone crisis, the FDA says — for cisgender men
December 12 2025 4:59 PM
Budapest mayor could face charges for hosting LGBTQ+ Pride march
December 12 2025 4:13 PM
Jason Collins, first out gay NBA player, reveals he has 'deadliest form of brain cancer'
December 12 2025 2:09 PM
The Democratic candidate in the Texas Senate race is going to be an LGBTQ+ ally
December 12 2025 12:55 PM
Texas expands lawsuits against doctors accused of providing gender-affirming care to youth
December 11 2025 4:36 PM
How Sundance 2026 celebrates its queer legacy
December 11 2025 3:54 PM
George Santos’s exclusive D.C. Christmas party featured famous grifters & MAGA influence peddlers
December 11 2025 3:31 PM
Nancy Mace investigated for bad behavior at airport, blames transgender people
December 11 2025 1:11 PM
Pete Buttigieg mocks Trump Transportation Secretary Sean Duffy’s strange airport pull-up stunt
December 11 2025 1:00 PM
Appeals court mulls upholding ruling that struck down Pentagon’s HIV enlistment ban
December 11 2025 11:51 AM
Florida sues leading medical groups for supporting gender-affirming care
December 11 2025 11:02 AM
Behind Marjorie Taylor Greene's latest push to criminalize gender-affirming care
December 10 2025 9:09 PM
Queer actor Wenne Alton Davis, known for 'Maisel,' 'Normal Heart,' killed in NYC car crash
December 10 2025 5:14 PM
‘Proud’ pro-LGBTQ+ Democrat flips Republican state House seat in Georgia electoral upset
December 10 2025 4:05 PM
Texas city votes to overturn LGBTQ+ antidiscrimination protections
December 10 2025 4:03 PM
Pornhub's spicy stats prove just how horny 2025 was
December 10 2025 3:30 PM
'Heated Rivalry' stars thank WeHo gay bar for 'tweeting about our butts'
December 10 2025 2:55 PM









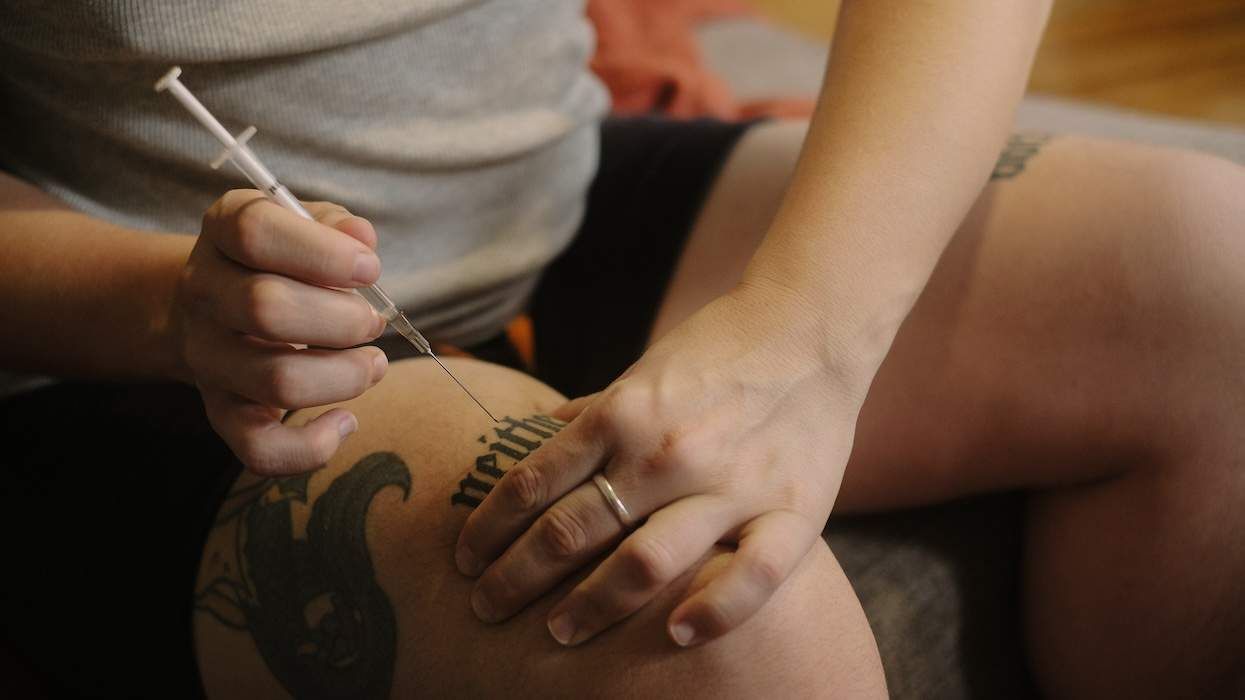
























































Charlie Kirk DID say stoning gay people was the 'perfect law' — and these other heinous quotes