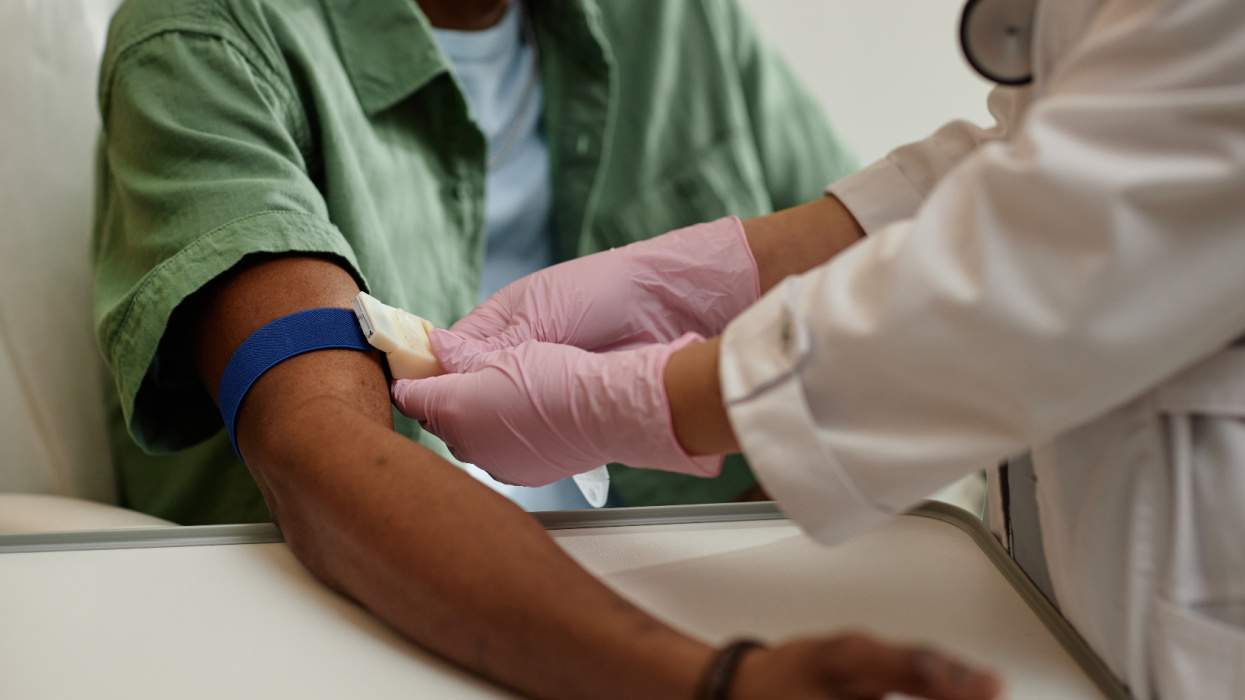
The Food and Drug Administration will begin a new fast-track review and approval process for fixed-dose pills containing two or more anti-HIV medications that can be shipped to poor nations as part of the President's Emergency Plan For AIDS Relief, USA Today reports. The fast-track process was unveiled by Health and Human Services secretary Tommy Thompson at a health conference in Geneva, Switzerland. The expedited process is intended to encourage the production of pills that contain two or more anti-HIV drugs to simplify drug-dosing requirements in developing countries where HIV-positive residents may have a more difficult time adhering to complicated treatment regimens. The expedited review process will apply to new products that combine drugs already FDA approved into a fixed-dose pill and to new copackaging of existing treatments. Generic drugs will also be more quickly reviewed, Thompson said, but these medications will have to be shown to meet FDA standards for safety and efficacy during the approval process, both individually and when combined with other anti-HIV drugs. Applications for fixed-dose combination pills could be approved in as little as two to six weeks under the new review guidelines. The expedited review process also simplifies the gathering and submission of drug-related data for FDA review, which under previous guidelines could take up to four years to produce. The new guidelines took effect immediately. The FDA on Monday published a new handbook for drug companies that includes information on how to apply for expedited review. "We are clearing the way to quickly deliver quality, life-saving HIV/AIDS drugs to people who desperately need them in developing countries," said Thompson in announcing the new review process. But many AIDS activists say the approval process for generic medications is still too lengthy, and actually delays getting generic drugs already studied and approved by the World Health Organization to countries hard-hit by AIDS. "While this sounds like a fast track, it's a delay," Paul Zeitz, executive director of the Global AIDS Alliance, told The Wall Street Journal. William Haddad, a spokesperson for the Indian generic drug firm Cipla, also said the review process was "unnecessary" and "unwarranted" for medications already approved by WHO.
Search
AI Powered
Human content,
AI powered search.
Latest Stories
Stay up to date with the latest in LGBTQ+ news with The Advocate’s email newsletter, in your inbox five days a week.
@ 2026 Equal Entertainment LLC.
All rights reserved
All rights reserved
By continuing to use our site, you agree to our Privacy Policy and Terms of Use.
The Latest
Support Independent Journalism
LGBTQ+ stories deserve to betold.
Your membership powers The Advocate's original reporting—stories that inform, protect, and celebrate our community.
Become a Member
FOR AS LITTLE AS $5. CANCEL ANYTIME.
More For You
Most Popular
@ 2026 Equal Entertainment LLC. All Rights reserved