CONTACTAbout UsCAREER OPPORTUNITIESADVERTISE WITH USPRIVACY POLICYPRIVACY PREFERENCESTERMS OF USELEGAL NOTICE
© 2025 Equal Entertainment LLC.
All Rights reserved
All Rights reserved
By continuing to use our site, you agree to our Privacy Policy and Terms of Use.
We need your help
Your support makes The Advocate's original LGBTQ+ reporting possible. Become a member today to help us continue this work.
Your support makes The Advocate's original LGBTQ+ reporting possible. Become a member today to help us continue this work.
The family of a pregnant woman who died while taking experimental anti-HIV drugs to protect her baby from getting the disease is suing the doctors, drugmakers, and hospitals involved in the study for $10 million. Joyce Ann Hafford's mother and sister allege in the suit filed Tuesday that doctors continued to give Hafford the drug regimen despite signs of liver failure. The suit also claims doctors didn't warn the 33-year-old HIV-positive woman of the trial's dangers. "She trusted doctors to treat her, and they failed her," said Rubbie King, Hafford's sister. A spokeswoman for Regional Medical Center in Memphis, Tenn., where Hafford was treated, said hospital policy prevented officials there from commenting on the suit. Family members said they did not learn that the National Institutes of Health had concluded the drug therapy most likely caused Hafford's death until the Associated Press obtained copies of the case file this month. For the past year, family members believed Hafford died of AIDS complications. NIH officials quickly suspected the drug regimen because it included Viramune (nevirapine), an antiretroviral drug known to cause liver problems. Hafford's death in August 2003, less than 72 hours after her son Sterling was born prematurely, halted the federal government research program on nevirapine. The baby was born HIV-negative. The family first filed a wrongful-death suit in June but withdrew it in September because lawyers felt they didn't have enough evidence. Among the defendants named in the suit are several doctors and nurses who treated Hafford, the Regional Medical Center, and drugmakers GlaxoSmithKline and Boehringer Ingelheim Pharmaceuticals. (AP)
From our Sponsors
Most Popular
Bizarre Epstein files reference to Trump, Putin, and oral sex with ‘Bubba’ draws scrutiny in Congress
November 14 2025 4:08 PM
True
Jeffrey Epstein’s brother says the ‘Bubba’ mentioned in Trump oral sex email is not Bill Clinton
November 16 2025 9:15 AM
True
Watch Now: Pride Today
Latest Stories
Rachel Maddow on standing up to government lies and her Walter Cronkite Award
December 15 2025 3:53 PM
Beloved gay 'General Hospital' star Anthony Geary dies at age 78
December 15 2025 2:07 PM
Rob Reiner deserves a place in queer TV history for Mike 'Meathead' Stivic in 'All in the Family'
December 15 2025 1:30 PM
Culver City elects first out gay mayor — and Elphaba helped celebrate
December 15 2025 1:08 PM
Texas city cancels 2026 Pride after local council rescinds LGBTQ+ protections
December 15 2025 12:55 PM
North Carolina county dissolves library board for refusing to toss book about a trans kid
December 15 2025 11:45 AM
Florida and Texas launch 'legal attack' in push to restrict abortion medication nationally
December 15 2025 11:18 AM
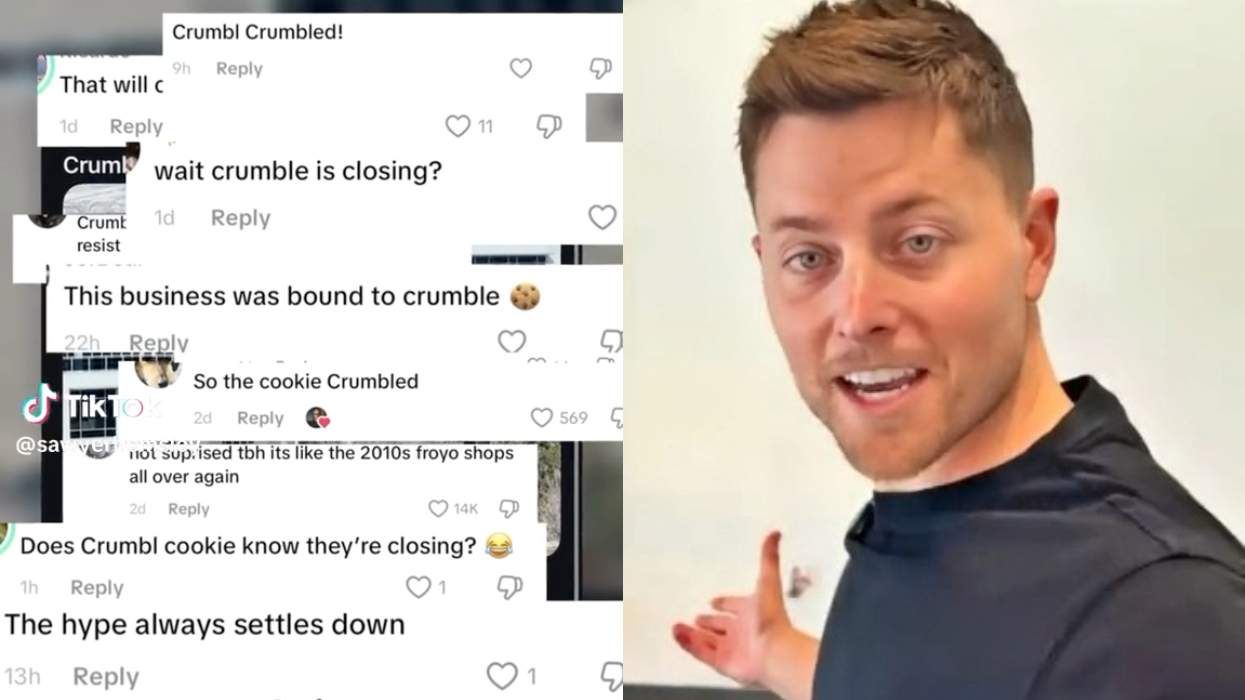
No, Crumbl is not Crumbl-ing, gay CEO Sawyer Hemsley says
December 15 2025 10:12 AM
11 times Donald Trump has randomly brought up his ‘transgender for everybody’ obsession
December 15 2025 9:22 AM
The story queer survivors aren't allowed to tell
December 15 2025 6:00 AM
Rob Reiner, filmmaker and marriage equality advocate, and wife Michele dead in apparent homicide
December 15 2025 1:08 AM
Trending stories
Recommended Stories for You
































































Charlie Kirk DID say stoning gay people was the 'perfect law' — and these other heinous quotes