October 25 2006 3:59 PM EST
CONTACTAbout UsCAREER OPPORTUNITIESADVERTISE WITH USPRIVACY POLICYPRIVACY PREFERENCESTERMS OF USELEGAL NOTICE
© 2025 Equal Entertainment LLC.
All Rights reserved
All Rights reserved
By continuing to use our site, you agree to our Privacy Policy and Terms of Use.
We need your help
Your support makes The Advocate's original LGBTQ+ reporting possible. Become a member today to help us continue this work.
Your support makes The Advocate's original LGBTQ+ reporting possible. Become a member today to help us continue this work.
The tens of thousands of people in the United States who are infected by hepatitis B each year were given access to a new drug on Wednesday to fight the virus.
The Food and Drug Administration said it approved Tyzeka, known generically as telbivudine, for use in treating adults with chronic hepatitis B. The drug is not a cure but can suppress the hepatitis B virus and reduce the liver inflammation that accompanies the disease, the FDA said.
Hepatitis B is caused by a virus that infects the liver. It can cause scarring, cancer, and failure of the liver as well as death. There are now six FDA-approved drugs to treat hepatitis B.
Tyzeka was developed by Idenix Pharmaceuticals Inc. of Cambridge, Mass., and the Swiss drug company Novartis Pharma AG.
The Hepatitis B Foundation estimates that nearly 100,000 people in the United States are infected annually. Most recover from the infection, although some go on to develop chronic conditions. About 5,000 people in the United States die each year from hepatitis B and its complications.
The virus can be spread through infected blood, typically during unprotected sex, childbirth, or through the sharing or reuse of hypodermic needles.
Side effects of Tyzeka include elevated levels of an enzyme suggestive of the breakdown of muscle tissue, upper respiratory tract infection, fatigue, headache, abdominal pain, and cough, the FDA said.
Among other drugs in the same class, there have been reports of fatal cases in which patients suffered severe enlargement and accumulation of fat in the liver, the FDA said. (AP)
From our Sponsors
Most Popular
Bizarre Epstein files reference to Trump, Putin, and oral sex with ‘Bubba’ draws scrutiny in Congress
November 14 2025 4:08 PM
True
Jeffrey Epstein’s brother says the ‘Bubba’ mentioned in Trump oral sex email is not Bill Clinton
November 16 2025 9:15 AM
True
Watch Now: Pride Today
Latest Stories
Transgender Army vet running for state delegate in red Maryland district is all about showing up
December 03 2025 7:00 AM
7 times Pete Hegseth was the definition of toxic masculinity
December 02 2025 5:46 PM
Man pleads guilty to murder of gay University of Mississippi student Jimmie 'Jay' Lee
December 02 2025 2:32 PM
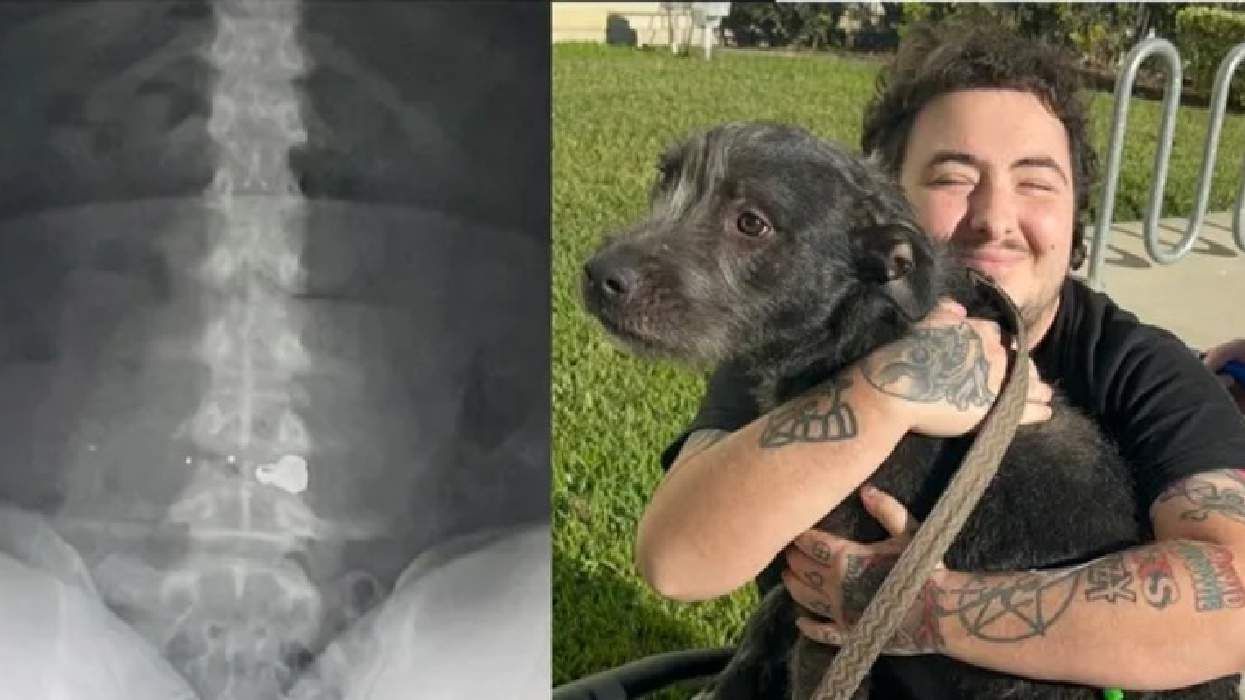
Florida man partially paralyzed after neighbor allegedly shot him and used anti-LGBTQ+ slurs
December 02 2025 1:30 PM
Queer comedian Cameron Esposito has first baby with wife Katy Nishimoto
December 02 2025 12:49 PM
Trans National Guard employee in Illinois sues Trump over restroom ban
December 02 2025 11:59 AM
Oklahoma University instructor suspended for failing student’s unscientific anti-trans psychology essay
December 02 2025 11:03 AM
Here are all of Trump's political enemies that have been charged or investigated (so far)
December 02 2025 9:52 AM
Joe Biden to receive top honor at LGBTQ+ leadership conference for his contributions to equality
December 02 2025 6:00 AM
On World AIDS Day, thinking of progress and how to build on it in the face of hostility
December 01 2025 7:47 PM
Ex-Biden White House aide called out for implying Cory Booker’s new marriage is suspicious
December 01 2025 6:04 PM
True
Trending stories
Recommended Stories for You


































































Charlie Kirk DID say stoning gay people was the 'perfect law' — and these other heinous quotes