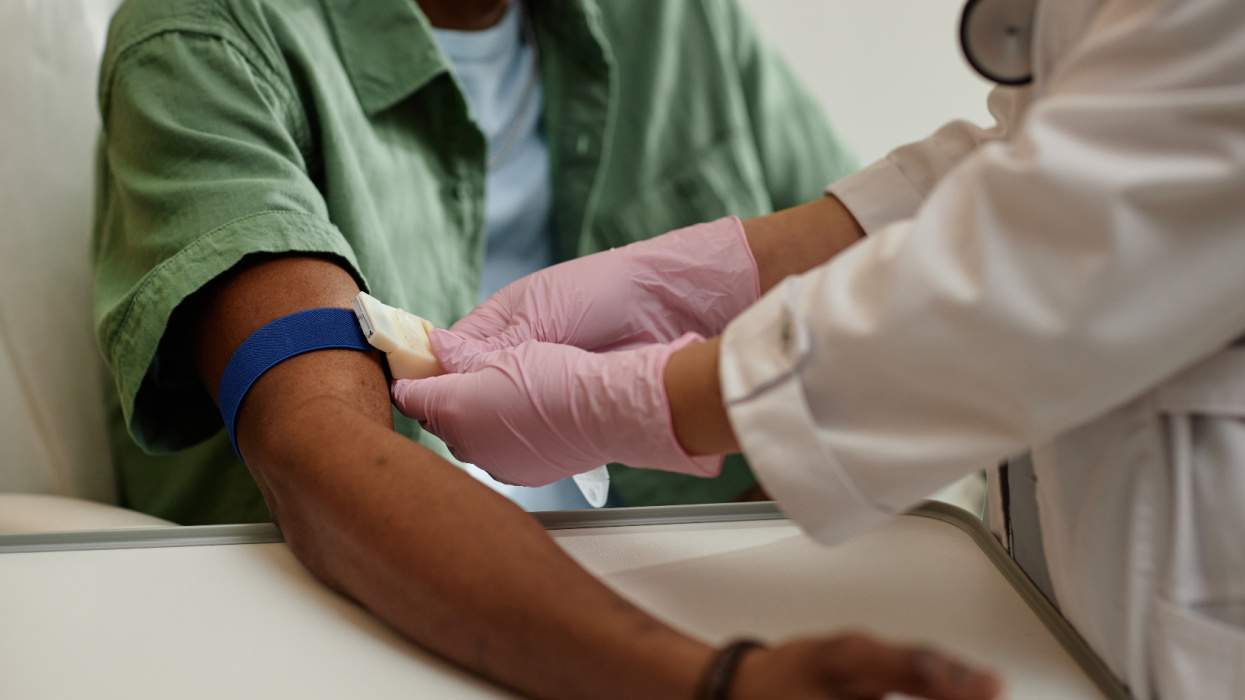
HIV infections have decreased dramatically among gay and bisexual men in the United Kingdom -- and researchers say PrEP is to thank.
New numbers released by the Public Health England show that rates have fallen by 71 percent in the last decade among this demographic, reports the BBC. There were 2,800 reported cases of HIV transmission in 2012, compared to 800 in 2018.
PrEP, or pre-exposure prophylaxis, is a treatment that studies have shown to be up to 99 percent effective in preventing HIV transmission if administered daily. In addition to PrEP, experts credit a rise in HIV testing and treatment as factors in the drop.
In 2018, 97 percent of the 103,800 people living with HIV were on treatment, making them undetectable for the virus and thus unable to pass it on to others.
The U.K. aims to end HIV transmission by 2030, and these numbers put the country on track to reach that goal. "HIV has brought untold hurt and suffering to so many, so it is encouraging to see transmissions continue to fall across the U.K.," Matt Hancock, Britain's secretary of State for Health and Social Care, told the BBC.
At present, PrEP is free to at-risk groups like the LGBTQ community in Scotland and Wales. However, the treatment is currently not widely available in England. This has been criticized by activists, who are calling on the government to fund PrEP for those who need it.
"The figures show the impact that PrEP, combined with increases in HIV testing and rapid access to HIV treatments can have," PrEP advocate Phil Samba told the BBC. "We truly are living in a new era of HIV prevention. Yet PrEP is still not freely available on the [National Health Service] in England, and unnecessary HIV infections are happening because of foot-dragging by politicians."
In the United States, PrEP is not widely available due to its high price. Truvada and Descovy are the only drugs approved in the U.S. for use as PrEP. Although cost supplement and donation programs exist, the manufacturer of the drugs, Gilead, is currently embroiled in a lawsuit with the federal government over the Truvada patent.