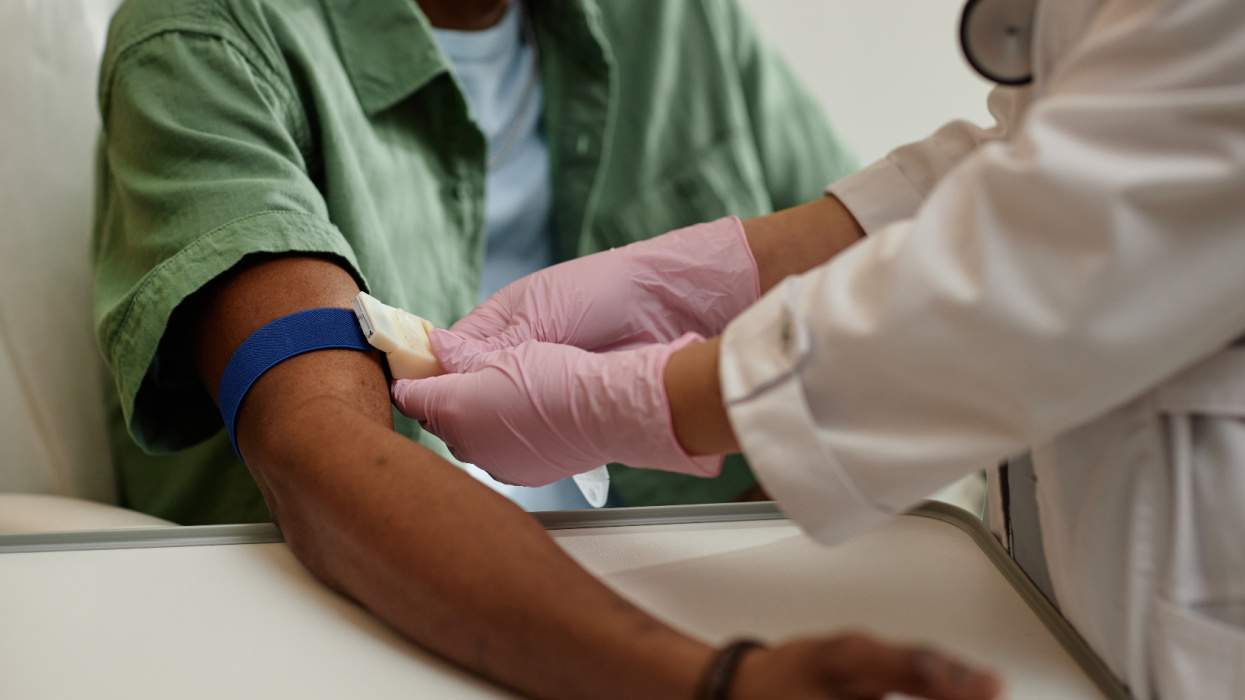
A three-year clinical trial evaluating the effectiveness of the anti-HIV drug Viread in preventing HIV infections among high-risk groups is set to begin next month in Thailand, the Bangkok Post reports. The clinical trial, funded by the U.S. Centers for Disease Control and Prevention and approved by Thailand's health ministry, will include about 1,600 volunteers, including injection-drug users and others deemed at high risk for HIV infection. Researchers will evaluate whether daily doses of Viread are effective in preventing HIV infections among the study subjects. Similar clinical trials are under way in the United States, Ghana, Cameroon, Nigeria, and Malawi. The U.S. trials include sexually active gay and bisexual men in San Francisco and Atlanta. The Cameroon trial was temporarily suspended last month after AIDS activists claimed that the female sex workers recruited for the study were not being given adequate HIV prevention counseling and did not fully understand the consent forms they signed for the study. After being reassured by trial funders that the trial adhered to ethical standards, Cameroon health officials agreed to allow it to continue. A similar clinic trial involving Cambodian sex workers was canceled by Prime Minister Hun Sen in August 2004 because of the possible effects of the drug on trial participants.
Search
AI Powered
Human content,
AI powered search.
Latest Stories
Stay up to date with the latest in LGBTQ+ news with The Advocate’s email newsletter, in your inbox five days a week.
@ 2026 Equal Entertainment LLC.
All rights reserved
All rights reserved
By continuing to use our site, you agree to our Privacy Policy and Terms of Use.
The Latest
Support Independent Journalism
LGBTQ+ stories deserve to betold.
Your membership powers The Advocate's original reporting—stories that inform, protect, and celebrate our community.
Become a Member
FOR AS LITTLE AS $5. CANCEL ANYTIME.
More For You
Most Popular
@ 2026 Equal Entertainment LLC. All Rights reserved