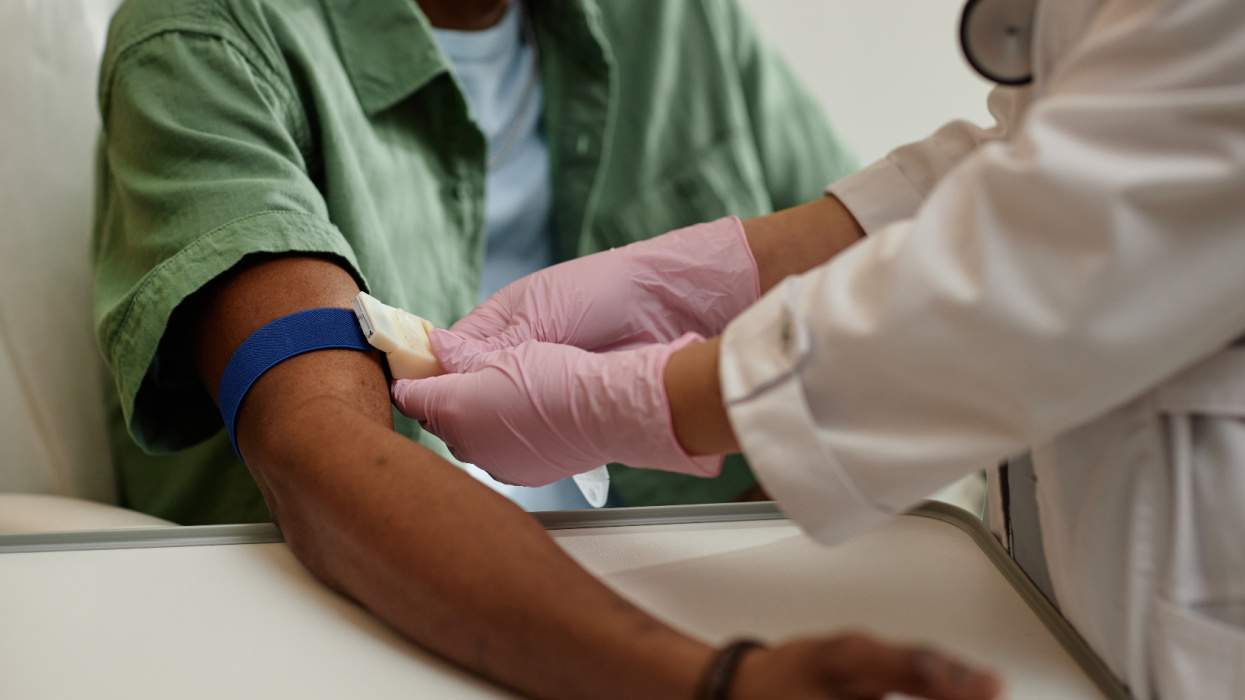
Pfizer's test of its CCR5 inhibitor drug, maraviroc, should be redesigned or otherwise stopped because it is unethically putting HIV-positive people at risk of developing AIDS, says the European AIDS Treatment Group. EATG criticizes Pfizer for allowing previously untreated people with HIV--regardless of their immune system health or viral load--to enroll in the Phase IIb/III trial of its CCR5 inhibitor drug. That could mean people with severely compromised immune systems who are at risk of developing AIDS would get an untested investigational drug rather than the proven anti-HIV drugs they need, says EATG, an influential voice in European AIDS policy. "The trial design should be changed or otherwise stopped," says Mauro Guarinieri, EATG's chairman. "We demand all concerned regulatory authorities to assume their responsibility and act accordingly." "Pfizer believes a thorough evaluation of maraviroc is required in a population representative of individuals initiating HIV therapy today," the company says. The maraviroc study has appropriate checks and balances to safeguard patient safety and, simultaneously, allows researchers to ascertain the drug's best future use, says Pfizer. Study design concerns have forced Pfizer to suspend planned tests of maraviroc in France, Germany, and Spain. However, the trials have been approved or are enrolling patients in Australia, Belgium, Canada, Italy, Mexico, Netherlands, Switzerland, the United Kingdom, and the United States. Pfizer is a few months ahead of drugmakers GlaxoSmithKline and Shering-Plough in its development of a drug to block HIV from entering healthy immune cells' CCR5 coreceptors, say industry analysts. The CCR5 drugs work differently than the injectable entry inhibitor Fuzeon and are in tablet form. The first CCR5 inhibitor could reach the market in 2007 or 2008, says Graeme Moyle, an HIV expert in London's Chelsea and Westminster Hospital. (Reuters)
Search
AI Powered
Human content,
AI powered search.
Latest Stories
Stay up to date with the latest in LGBTQ+ news with The Advocate’s email newsletter, in your inbox five days a week.
@ 2026 Equal Entertainment LLC.
All rights reserved
All rights reserved
By continuing to use our site, you agree to our Privacy Policy and Terms of Use.
The Latest
Support Independent Journalism
LGBTQ+ stories deserve to betold.
Your membership powers The Advocate's original reporting—stories that inform, protect, and celebrate our community.
Become a Member
FOR AS LITTLE AS $5. CANCEL ANYTIME.
More For You
Most Popular
@ 2026 Equal Entertainment LLC. All Rights reserved