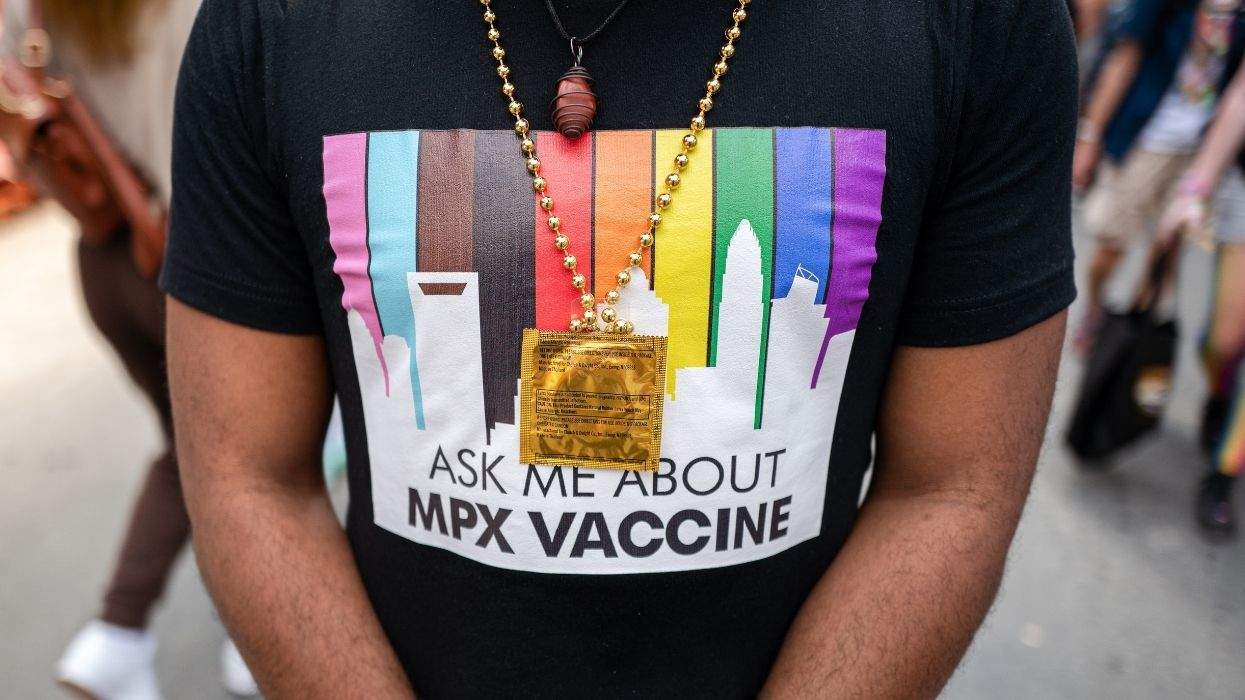
The Food and Drug Administration has announced that an inspection of Red Cross blood collection and distribution centers across the country has revealed more than 200 violations of federal blood safety laws, including being unable to account for blood samples that contained HIV and human cytomegalovirus, the Los Angeles Times reports. In a 45-page report, FDA inspectors say violations included lack of management control and quality assurance oversight, failure to correct violations from previous inspections, and the release of "unsuitable" blood products. The FDA also found evidence of employees falsifying records and shipping orders without carrying out blood testing, including blood labeled as "unsuitable" that was shipped despite the required testing. Red Cross officials say that many of the problems cited in the report have been corrected through a recent $280 million investment to improve the safety of the nation's blood supply, including hiring new staff and upgrading computer networks. "The Red Cross understands more work needs to be done to further strengthen our processes and procedures, and we are fully committed to working collaboratively with the FDA to enhance our system," said Ramesh Thadani, Red Cross executive vice president and CEO of biomedical services. FDA commissioner Mark McClellan noted that while the report shows that there is considerable room for improvement at the Red Cross, the FDA still considers the nation's blood supply to be safe. He also noted that there was no evidence to show that blood donors faced any health risks. Experts say the chance of getting HIV from donated blood is one in 2 million to 3 million transfusions.
Search
AI Powered
Human content,
AI powered search.
Latest Stories
Stay up to date with the latest in LGBTQ+ news with The Advocate’s email newsletter, in your inbox five days a week.
@ 2026 Equal Entertainment LLC.
All rights reserved
All rights reserved
By continuing to use our site, you agree to our Privacy Policy and Terms of Use.
The Latest
Support Independent Journalism
LGBTQ+ stories deserve to betold.
Your membership powers The Advocate's original reporting—stories that inform, protect, and celebrate our community.
Become a Member
FOR AS LITTLE AS $5. CANCEL ANYTIME.
More For You
Most Popular
@ 2026 Equal Entertainment LLC. All Rights reserved