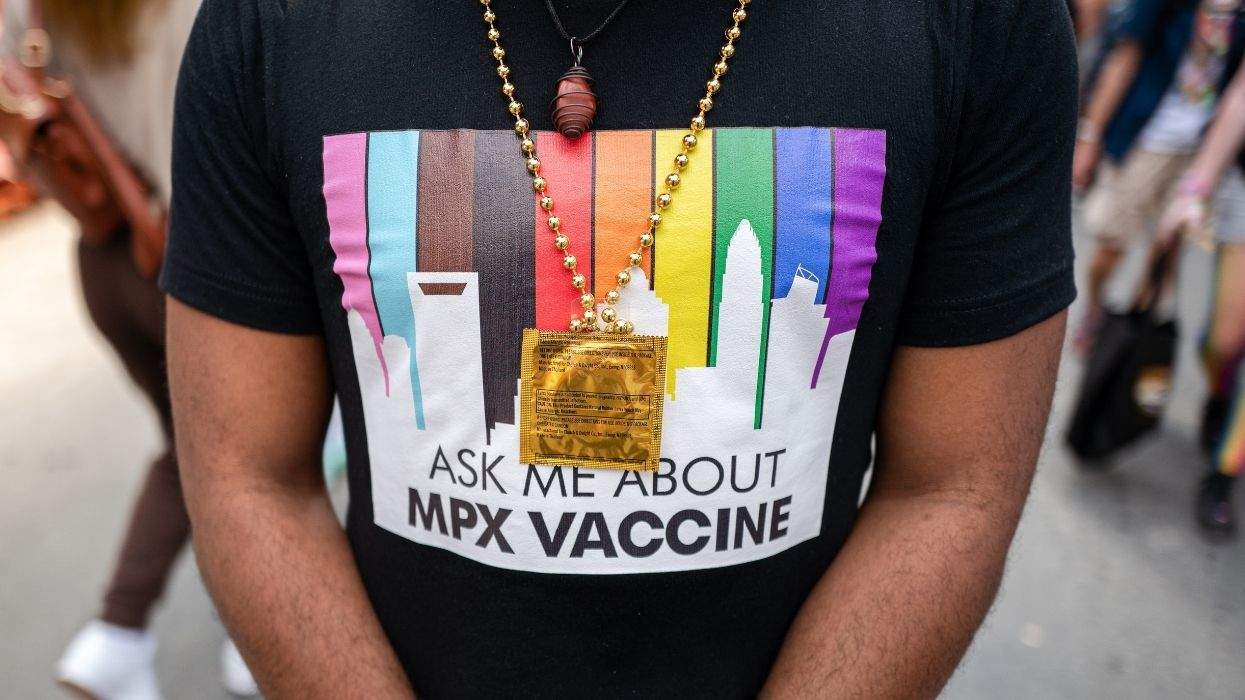
Pharmaceutical companies Merck and Aventis this week announced that they are combining their two most promising HIV vaccines for a joint human trial set to begin later this year, The Wall Street Journal reports. They decision followed an animal study that showed the two vaccines when administered together created a greater immune response than either vaccine alone. Neither company has been able to determine why the experimental vaccines work better in tandem than separately. Merck's vaccine is known as MRKAd5, and Aventis's vaccine is called ALVAC. Both vaccines aim to produce immune system responses against invading HIV to prevent infection. Company officials say they plan to begin human trials of the compounds later this year. They will announce the specific start date once the Food and Drug Administration approves their clinical trial application. Seth Berkley, president and CEO of the International Vaccine Initiative, praised the decision to test the vaccines together. "By joining forces, Merck and Aventis have made a bold move that could advance the world considerably closer to discovering an AIDS vaccine," he said. "A vaccine is the best hope to end the spread of a virus that infects nearly 15,000 men, women and children daily and threatens the survival of whole communities."
Search
AI Powered
Human content,
AI powered search.
Latest Stories
Stay up to date with the latest in LGBTQ+ news with The Advocate’s email newsletter, in your inbox five days a week.
@ 2026 Equal Entertainment LLC.
All rights reserved
All rights reserved
By continuing to use our site, you agree to our Privacy Policy and Terms of Use.
The Latest
Support Independent Journalism
LGBTQ+ stories deserve to betold.
Your membership powers The Advocate's original reporting—stories that inform, protect, and celebrate our community.
Become a Member
FOR AS LITTLE AS $5. CANCEL ANYTIME.
More For You
Most Popular
@ 2026 Equal Entertainment LLC. All Rights reserved