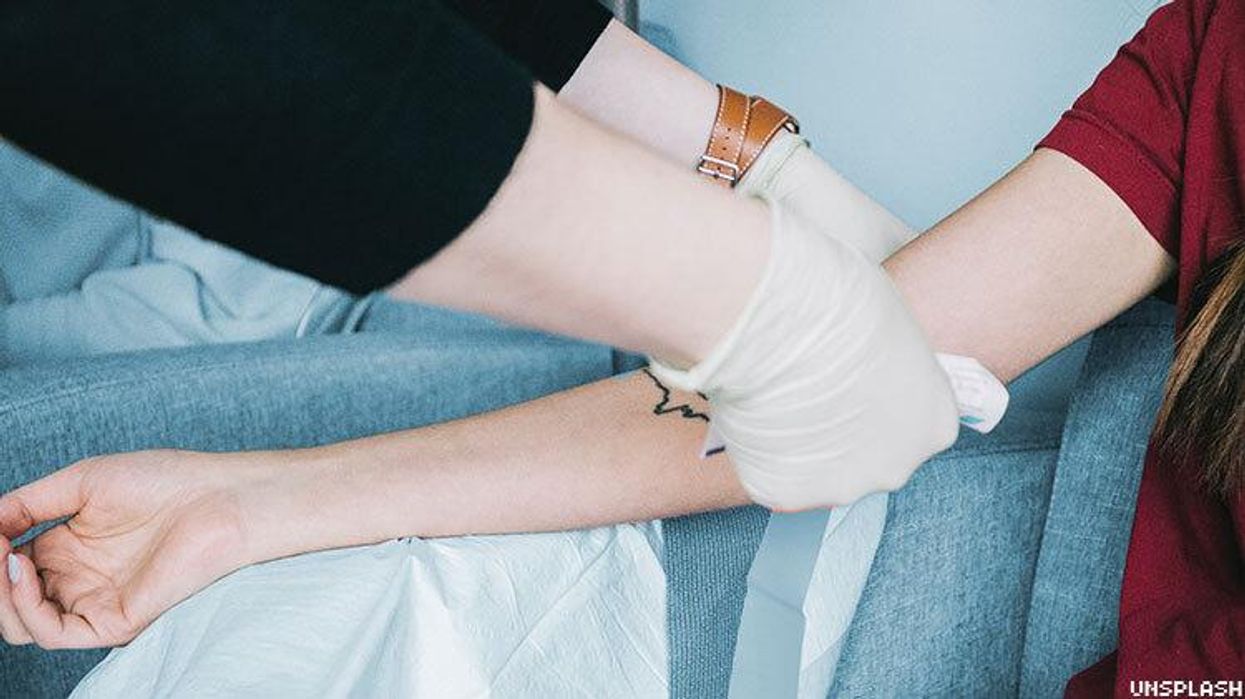
Attorneys general from 19 states and the District of Columbia are urging the federal government to change its policy on blood donations by gay and bisexual men, moving to guidelines based on individual risk factors rather than restricting entire groups of people.
In early April, the Food and Drug Administration put out new guidance for U.S. blood centers saying men should be allowed to donate if they have not had sex with another man in the past three months. This is down from a year, under a policy adopted in 2015, and a lifetime ban on donations by men who have sex with men, adopted in the 1980s, at the height of the AIDS epidemic and before there was a reliable way to screen blood for HIV. Blood centers do remain free to enforce more restrictive policies.
The new guidance is a step in the right direction but doesn't go far enough, say the attorneys general, led by California's Xavier Becerra. It remains discriminatory and does not reflect current science, plus there is a shortage of blood and its components due to the COVID-19 pandemic, with the cancellation of blood drives and drop in donations.
"As Americans stay home to stop the spread of coronavirus, the nation is facing a shortage of blood donations, which provide critical medical support to hospitals and their patients," Becerra said in a press release. "During this pandemic, it is important to continue to evaluate and modernize blood donation guidance to be inclusive of LGBTQ Americans. A risk-based model not only protects the health and safety of our communities -- it's the right thing to do."
"Ensuring that the blood supply is safe is an important goal and donor blood is extensively tested for infectious disease pathogens before a blood transfusion," reads the letter, addressed to Adm. Brett Giroir, the assistant secretary for health at HHS. "Additional precautions regarding who can donate blood should be narrowly tailored to achieve safety goals while maximizing the blood supply. The revised guidance still precludes many LGBTQ Americans from fully contributing to the blood shortages while still requiring a waiting period for healthy individuals. Further, the FDA should immediately clarify that the new policies do not bar MSM from donating potentially lifesaving convalescent plasma to their loved ones."
It notes, for instance, that "strict application of this policy could still bar a gay man who recovers from COVID-19 from donating convalescent plasma to his critically ill husband, even if they are both HIV-negative and even if their relationship is exclusively monogamous. ... The FDA should immediately clarify that the new three month MSM deferral period announced does not bar MSM from donating convalescent plasma to their loved ones."
Some countries, the letter points out, have maintained the safety of their blood supply with a gender-neutral policy that screens out donors who have multiple sexual partners or recurrent sex with a partner whose HIV status is unknown.
Lifting the ban would greatly increase the blood supply, it says, citing a Williams Institute study concluding that "lifting the blood donation ban among MSM could be used to help save the lives of more than a million people."
"The FDA's revised policy will help address the current shortage of blood and blood products during these unprecedented times," the letter asserts. "But transitioning to a risk-based model will further protect the blood supply and donor's dignity."
The attorneys general join physicians in calling for further changes to the policy. More than 500 doctors signed on to a letter to the FDA, released last week, calling for "scientifically driven standards that uphold the utmost safety of the blood supply and simultaneously promote equity and reverse historical discrimination in blood donation." They noted there are sophisticated means of screening blood for HIV and that men who are in monogamous same-sex relationships or who are taking pre-exposure prophylaxis are at low risk for the virus.